 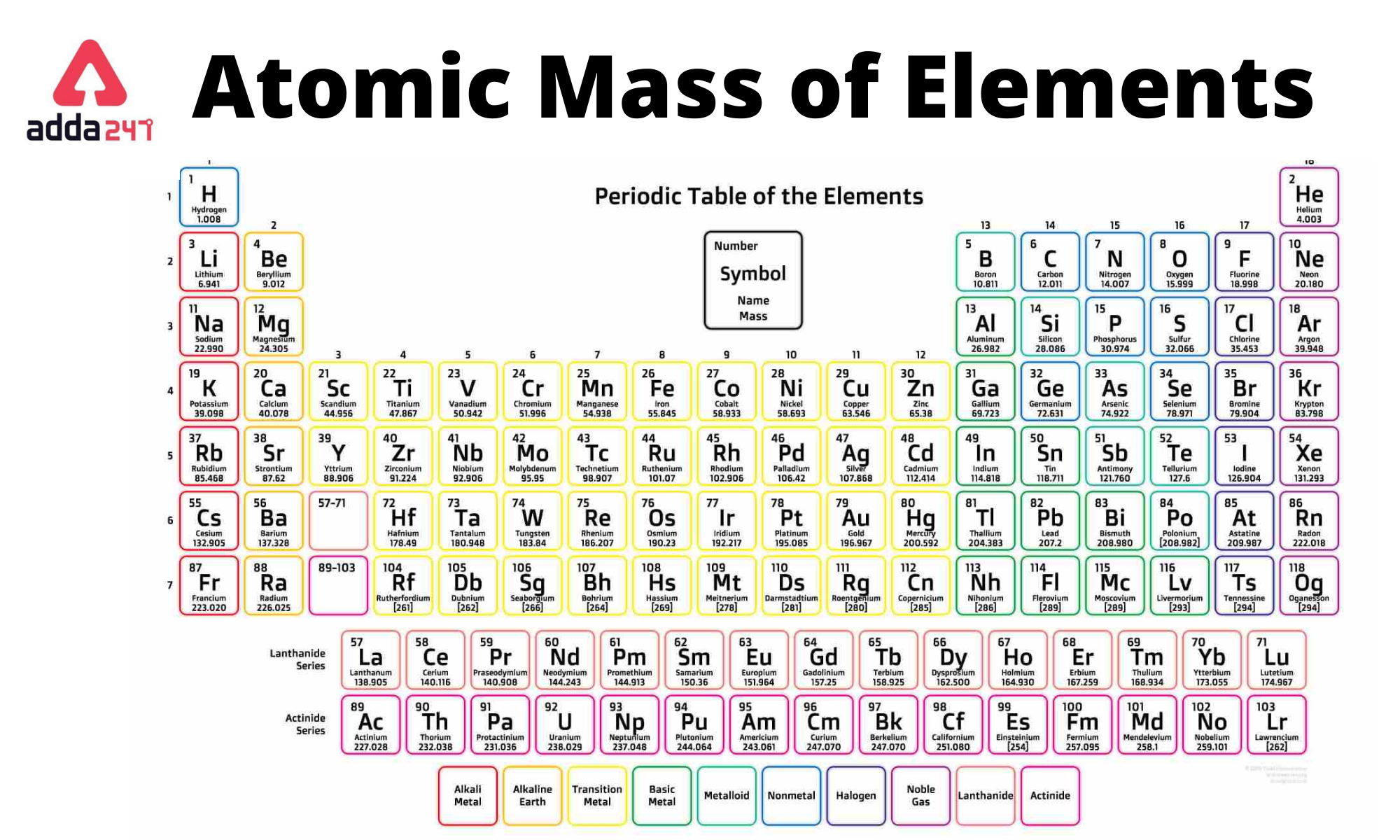
124.3 L PCls(g) convert to mol PCls(g) at STP (1 atm and 273. 124.3 L PCls(g) convert to mol PCls(g) at STP (1 atm and 273.15 K)ģ. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in C2Cl4F2: Molar Mass (g/mol) C (Carbon) 2 × 12.0107 24.0214. 2.6 μmol KCO3 convert to mass KCO3 (Molar Mass KCO3 = 138.21 g/mol)ģ. 8.32x10^2 molecules CH4 convert to mol CH4Ģ. Show a T-Chart for all problems, using the molar mass given if needed.ġ. (Example: Carbon 12.0g)įor the following measurements, convert to the requested unit. Round all answers to two digits after the decimal. Use the periodic table linked in Google Classroom for the molar masses. For the following chemical compounds and molecules, calculate the molar mass. The molar mass of carbon dioxide is 12.01 + (2 × 16.00) = 44.01 g/mol.Solve the following problems, showing all work and units for all problems. 
CO 2 has one carbon atom and two oxygen atoms.Oxygen (O) has an atomic mass of about 16.00 amu.Carbon (C) has an atomic mass of about 12.01 amu.Let's calculate the molar mass of carbon dioxide (CO 2): Add them together: add the results from step 3 to get the total molar mass of the compound.The molar mass is calculated in two ways. Avogadros number tells the number of particles present per mole. C Add together the masses to give the molecular mass. The periodic table provides the atomic masses used in the calculation of the molar mass. Calculate molar mass of each element: multiply the atomic mass of each element by the number of atoms of that element in the compound. B Obtain the atomic masses of each element from the periodic table and multiply the atomic mass of each element by the number of atoms of that element.The atomic mass is usually found on the periodic table and is given in atomic mass units (amu). 
Now here we have to find the molar mass of C2H2 (Ethyne). Because the molar mass of any molecule (or compound) can be calculated by simply adding the molar masses of individual atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
